 |
 |


|
|
|
| Intermediate of Cefozopran(CAS:197897-11-3) |
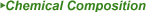 |
| 1-[((7R)-7-amino-4-carboxy-3,4-didehydrochepham-3-yl)methyl]-1H-imidazo[1,2-b] pyridazin-4-ium Iodide |
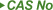 |
| 197897-11-3 |
 |
| Intermediate of Cefozopran |
 |
Cefozopran HCl and the Intermediates:
(1) Imidazo[1,2-b]pyridazine
CAS: 766-55-2
Standard: Enterprise Standard
Package: 20kg/25kg
(2) 1-[((7R)-7-amino-4-carboxy-3,4-didehydrochepham-3-yl)methyl]-1H-imidazo[1,2-b] pyridazin-4-ium Iodide
CAS: 197897-11-3
Standard: Enterprise Standard
Package: 20kg/25kg
(3)
(Z)-2-(5-amino-1,2,4-thiadiazol-3-yl)-2-(methoxyimino)acetic acid
CAS: 72217-12-0
Standard: Enterprise Standard
Package: 20kg/25kg
(4)
S-2-benzothiazolyl (Z)-2-(5-amino-1,2,4-thiadiazol-3-yl)-2-methoxyimino thioacetate
CAS: 89604-91-1
Standard: Enterprise Standard
Package: 20kg/25kg
(5) Cefozopran
CAS : 113359-04-9
Standard: Enterprise Standard
Package: 20kg/25kg
(6) Cefozopran hydrochloride
CAS :113981-44-5
Standard: JP15
Package: 5kg/10kg
(7) Mixture of Cefozopran HCl and Sodium carbonate
Standard: Enterprise Standard
Appearance: White to pale yellow, crystals or crystalline powder
Use: to produce Cefozopran HCl for injection.
|
|
|
|
|
|